Quantify enrollment risk before launch with real patients—
not assumptions.




The "Incentive" Gap
Investigators are incentivized to be optimistic to win the study. CROs are incentivized to be optimistic to win the bid. This creates a "Hope-Based" model where everyone agrees on an estimate that has never been tested against a live patient.
The "Historical" Trap
Most feasibility reports are "look-backs"—historical data from patients who were treated years ago under different standard-of-care conditions. They don't account for the patient sitting in a doctor's office today who is already on a prohibited medication.
The "Unseen" Bottleneck
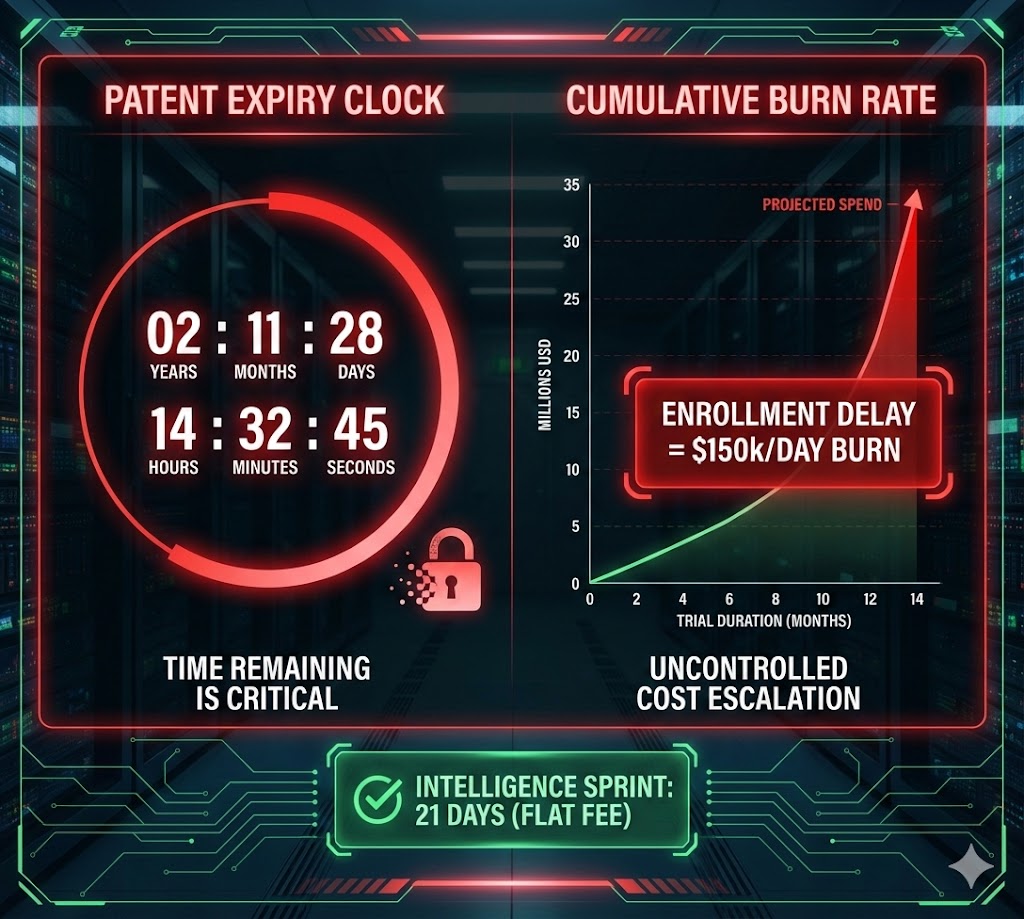
You can estimate site capacity, but you can't know the Screen-Fail Rate of a draft protocol until it hits the real world...after losing months of runway
Estimates are free. Delays cost millions.

De-risk your trials via real-time feasibility signals generated before the first site is activated.
We measure real patient interest & eligibility against proposed inclusion/exclusion criteria — prior to study launch.
We don’t ask investigators if they can find patients—we find them ourselves. Our data is independent of site optimism and CRO bidding pressure. You get an unvarnished, third-party audit of your protocol’s "enroll-ability" based on actual patient response, not a doctor’s best guess.
History is a poor predictor of current recruitment. Our Stress Test captures the real-world comorbidities and medication profiles of patients in the digital landscape today. We identify the disqualifying beta-blocker or the slightly-too-high BMI, in real-time.
We don’t just flag risks; we quantify the screen-fail. We provide the percentage drop-off for every I/E criteria. This allows the CMO to see exactly which "scientific" requirement is acting as a "recruitment" anchor, providing the data needed to optimize before the first site is ever activated.
$535,000 per Protocol Amendment
Months of enrollment delays
20-30% Protocol Failure Rates
$??MM in lost Patent Life
Discovery to delivery, we follow a proven framework that ensures clarity, precision, and growth.




Answers to common questions about our platform and services.
